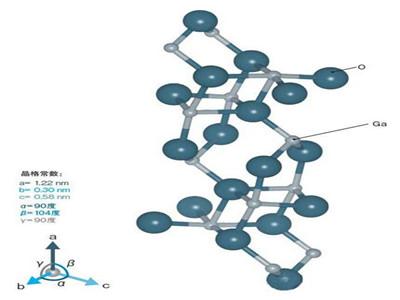
Gallium(III) trioxide (Ga2O3) 99.99%+ biraha raadraaca 12024-21-4
| Gallium oxide | |
| Kiimikada caanaha | Ga2O3 |
| cufka molar | 187.444 g/mol[1] |
| Muuqashada | budo crystalline cad |
| Cufnaanta | 6.44 g/cm3, alfa;5.88 g/cm3, beta |
| barta dhalaalaysa | 1,900°C (3,450°F; 2,170K) alfa;1725°C, beta |
| Biyo la'aanta | aan la milmin |
| Solubility | ku milma inta badan asiidhyada |
Tilmaamaha Gallium Oxide Tayada Sare
| Shayga Lam. | Halabuurka Kiimikada | Cabbirka Qaybaha | ||||||||
| Ca2O3 | Mat.(Natiijada Kormeerka) PPM | D50 | ||||||||
| (wt%) | Ni | Cu | Ca | Fe | Zn | In | Mn | Cr | μm | |
| UMGO4N | ≥99.99% | 8.2 | 3.8 | 2.5 | 2.4 | 0.8 | 0.4 | 0.1 | 0.1 | 3.82 |
Xirmooyinka: 25kg / dhalo caag ah, 20 dhalo / kartoon.
Maxaa Gallium Oxide loo isticmaalaa?
Gallium oxidewaxaa loo isticmaalaa laysarka, fosfooraska, iyo walxaha iftiinka.Monoclinic ß-Ga2O3 ayaa lagu dabaqay dareemayaasha gaaska, fosfoorrada luminescent iyo dahaarka dielectric ee unugyada qorraxda.Sida oksaydh deggan, waxa kale oo ay leedahay awood qoto dheer-ultraviolet hufnaanta conductive oxides, iyo codsiyada transistor.Filimada Ga2O3 ee khafiifka ah ayaa ah kuwa xiiseeya ganacsi ahaan sida walxaha xasaasiga u ah gaaska.ß-Gallium(III) oxide waxaa loo isticmaalaa soo saarista Ga2O3-Al2O3 kicinta.Sidoo kale la heli karo: Gallium Oxide sputtering Bartilmaameedyada Ga2O3.
Halkan ku qor fariintaada oo noo soo dir